Tag: salinity

Brine rejection and overturning, but not the way you think! Guest post by Robert Dellinger
It’s #KitchenOceanography season! For example in Prof. Tessa M Hill‘s class at UC Davis. Last week, her student Robert Dellinger posted a video of an overturning circulation on Twitter that got me super excited (not only because as of now, April 15th, it has 70 retweets and 309 likes. That’s orders of magnitude more successful than any […]
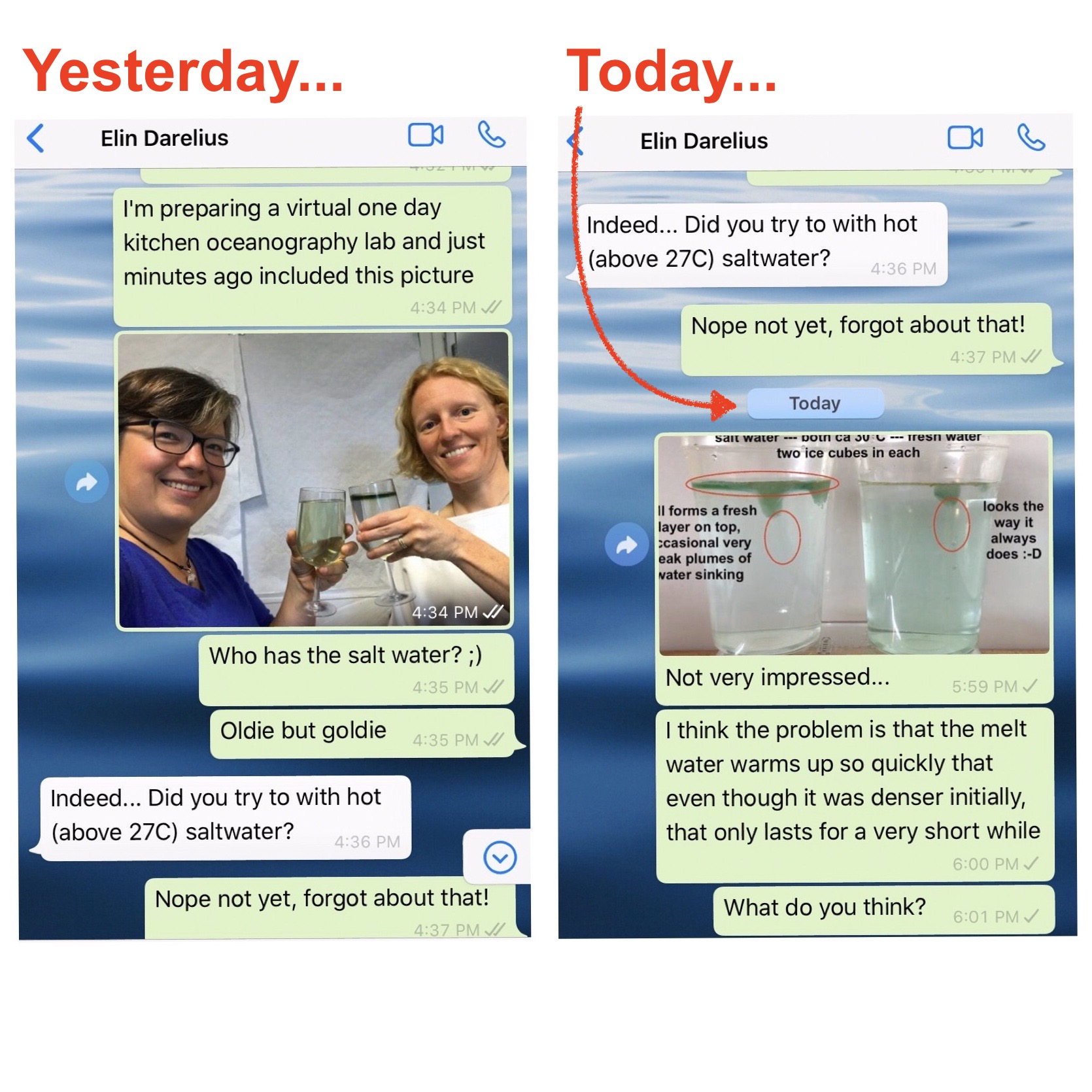
On melting ice cubes and molecular diffusion of heat
First of all, let me say how much I love having chats like the one Elin and I had over the weekend (which you only see the very beginning of above). I had gotten into a bit of a rut kitchen oceanography-wise, which, I am happy to report, is over now! Thanks, Elin! :-) One […]

Temperature dependency of molecular diffusion, and convection taking over
I saw the idea for this experiment on Instagram (Max is presenting it for @glaeserneslabor) and had to try it, too! The idea is to put drops of dye into hot and cold water and observe how in hot water the dye is mixed a lot faster than in cold water — after all, molecules […]

Ice cubes melting in fresh water and salt water
Today we are doing the melting ice cubes experiment in fancy glasses, because Elin is giving a fancy lecture tonight: The Nansen Memorial Lecture of the Norwegian Science Academy in Oslo! Cheers! We each had green ice cubes in our glasses, but one of our glasses contained fresh water and the other one salt water, […]

A “Siel” – the valve in a dyke that lets freshwater out but no salt water in
Ok now, after complaining about how I dislike mud and the “no water” (i.e. low water) times in the Wadden Sea yesterday, today I’ll tell you about some stuff I really love about the North Sea coast. For example, the pretty little villages full with shrimp trawlers everywhere! Picture above is Neuharlingersiel, below is Dornumersiel. […]

Demonstration: Nansen’s “dead water” in a tank!
A ship that is continuously pulled with a constant force suddenly slows down, stops, and then continues sailing as if nothing ever happened? What’s going on there? We will investigate this in a tank! And in order to see what is going on, we have dyed some of the water pink. Can you spot what […]

Accidental double-diffusive mixing
When setting up the stratification for the Nansen “dead water” demo (that we’ll show later today, and until then I am not allowed to share any videos, sorry!), I went into a meeting after filling in layer 4 (the then lowest). When I came back, I wanted to fill in layer 5 as the new […]

Guest post: Using seawater to make bread!
Last week I got one of the coolest emails I have ever received: Someone had found my blog while googling for the salt content of seawater in order to use it to make bread, and he sent me a couple of pictures the resulting bread! Of course, I asked if I could share it as […]

Experiment: Temperature-driven circulation
My favorite experiment. Quick and easy and very impressive way to illustrate the influence of temperature on water densities. This experiment is great if you want to talk about temperature influencing density. Although it doesn’t actually show anything different from a temperature driven overturning experiment, where circulation is determined by hot water rising and cold […]

Melting ice cubes experiment — observing the finer details
If you don’t know my favourite experiment for practically all purposes yet (Introduction to experimenting? Check! Thermohaline circulation? Check! Lab safety? Check! Scientific process? Check! And the list goes on and on…), check it out here. (Seriously, of you don’t recognize the experiment from the picture below, you need to read up on it, it’s […]

Evaporating sea water
How much salt is there in sea water? What concentration do you need before crystals start forming? What will those crystals look like? I am sure those are the kind of questions that keep you awake at night! Of course this can easily assessed experimentally. On a visit to the University of Bergen’s Centre for […]

Taking the hydrostatic paradox to the next (water) level
How well do people understand hydrostatics? I am preparing a workshop for tomorrow night and I am getting very bored by the questions that I have been using to introduce clickers for quite a lot of workshops now. So I decided to use the hydrostatic paradox this time around. The first question is the standard […]
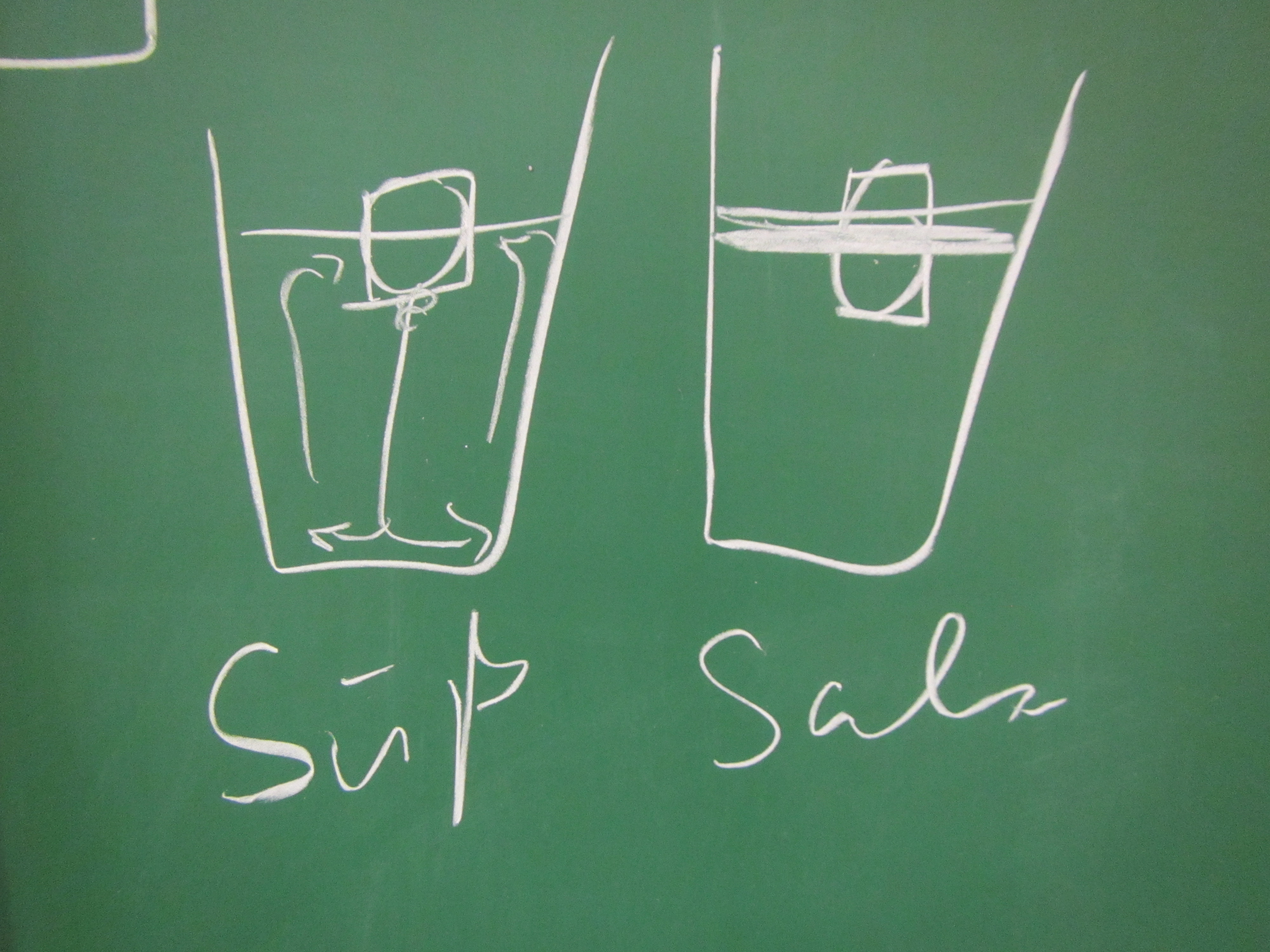
Using the “melting ice cube” experiment to let future instructors experience inquiry-based learning.
Using the “melting ice cube” experiment to let future instructors experience inquiry-based learning. I recently (well, last year, but you know…) got the chance to fill in for a colleague and teach part of a workshop that prepares teaching staff for using inquiry-based learning in their own teaching. My part was to bring in an experiment […]

When water doesn’t seek its level
Last week we talked about misconceptions related to hydrostatic pressure, and how water always seeks its level. Today I’m gonna show you circumstances in which this is actually not the case! We take the fat separator jug we used last week. Today, it is filled with fresh water, to which we add very salty water through the jug’s […]

My favorite demonstration of the coolest mixing process: Salt fingering!
I am updating many of my old posts on experiments and combining multiple posts on the same topic to come up with a state-of-the-art post, so you can always find the best materials on here. And today I would like to present you my favorite experiment: Salt fingering! Check out the new page I made for […]

Guest post: Estimating salinity as a homework assignment
Today I am super excited to share a guest post that my awesome friend Joke Lübbecke wrote for us. Joke is a professor in physical oceanography in Kiel, and we like to chat about teaching occasionally. She has great ideas for exciting tasks for students to do and I bet they learn a lot from her. […]

Ice cubes melting at the bottom of the beakers
Because surely there is one more post in this topic? ;-) For those of you who haven’t heard about the “melting ice cube” obsession of mine, please check out the links to other posts at the end of this post. For everybody else’s sake, let’s dive right in! When Kristin and I ran the workshop […]

Creating a continuous stratification in a tank, using the double bucket filling method
Because I am getting sick of stratifications not working out the way I planned them. Creating stratifications, especially continuous stratifications, is a pain. Since I wanted a nice stratification for an experiment recently, I finally decided to do a literature search on how the professionals create their stratifications. And the one method that was mentioned over […]

Double overflow
Because sometimes one overflow simply isn’t enough. Finn’s group came up with – and ran – an overflow experiment with many different densities and even more colors. While the movie didn’t turn out too well, the idea was pretty awesome. Rolf went ahead and modeled the experiment right away. And because the plume didn’t go across […]

Forced internal waves in a continuous stratification
Plus all kinds of dyes. (deutscher Text unten) Using the continuous salinity stratification created yesterday, Rolf and Daniel conducted a really cool experiment: They forced internal waves and watched them develop. I’ve converted their movie into a time-lapse; watch it below. Mit der kontinuierlichen Salzschichtung, die Daniel und Rolf gestern gebastelt haben, haben sie danach […]

Creating a continuous stratification.
And watching internal waves – a data-model comparison. (deutscher Text unten) In an experiment similar to the one done by the group looking at the effects of temperature and salinity on density, the wave group, supported by Rolf, started looking at how to create a continuous stratification through internal wave action. Two water masses, one saline […]

Effects of temperature and salinity on density and stratification
Removing a barrier between waters of different densities and watching what happens. (deutscher Text unten) Today, one of the groups performed a classical experiment (shown for example here) – but the awesome thing is that they came up with the planning pretty much by themselves in order to determine the effects of temperature and salinity […]

Guest post: The mystery of the cold room
Guest post by Kristin Richter! Today I’m excited to bring to you a guest post from Innsbruck, Austria, written by my friend Kristin Richter. Kristin ran the oceanography lab in Bergen before I took over, and she is a total enabler when it comes to deciding between playing with water, ice and food dye, or doing “real” […]

Help! Equation of State?
Is there an equation of state for hypersaline water at very cold temperatures? A friend of mine is looking to calculate changes in density of a hypersaline Antarctic lake from summer to winter. Apparently, this lake is about 10 times saltier than the ocean and often cools down to -17C at the bottom. My own spontaneous […]

Molecular diffusion of heat and salt
Why heat and salt diffuse at different rates. Why do heat and salt diffuse at different rates? This seems to always be puzzling students when talking about double diffusion. Well, why should they diffuse at the same rate? The processes of molecular diffusion of heat and salt are very different. In the case of heat, […]

Salt fingering – DIY
How to easily set up the stratification for the salt fingering process. Setting up stratifications in tanks is a pain. Of course there are sophisticated methods, but when you want to just quickly set something up in class (or in your own kitchen) you don’t necessarily want to go through the whole hassle of a […]

Diffusive layering. Or: This is not a trick question!
The “other” double-diffusive mixing process. After having talked extensively about double diffusive mixing in my courses, I tend to assume that students not only remember that there is such thing as double-diffusive mixing, but that they also remember our discussions on how the process works, and that they would be able to transfer this to processes […]

Salt fingering
How to show my favorite oceanographic process in class, and why. As I mentioned in this post, I have used double-diffusive mixing extensively in my teaching. For several reasons: Firstly, I think that the process is just really cool (watch the movie in this post and tell me that it isn’t!!!) and that the experiments […]

Double-diffusive mixing
On the coolest process in oceanography. My favorite oceanographic process, as all of my students and many of my acquaintances know, is double-diffusive mixing. Look at how awesome it is: Double-diffusive mixing happens because heat and salt’s molecular diffusion are very different: Heat diffuses about a factor 100 faster than salt. This can lead to […]

How sound is refracted towards the regions of minimum speed.
Students acting out the process of sound being refracted towards the region of minimum speed. We’ve been talking about refraction lately. Waves get bent in the direction of lower velocity. This holds for light and sound and even ocean waves. However, students find it conceptually difficult to understand why waves are being bent towards lower […]

Measuring salinity
Students evaporate water to measure the salinity of a water sample. As described in this post, I like to have students build “instruments” to measure the most oceanographic properties (temperature, salinity and density). I find that they appreciate oceanographic data much more once they have first-hand experience with how difficult it is to design instruments and […]

Tasting sea water reloaded
Doing the “tasting sea water” activity again with a different group of students. A very good introduction to the concept of salinity is the “tasting sea water” activity. Last time I ran that activity, students were very quick to correctly connect the samples with the correct sampling locations without much discussion going on. This time […]

How a CTD works
Movie on how the most important instrument in oceanography works. On our cruise on the WHOI research vessel Knorr in 2011, Sindre Skrede (find him on twitter or vimeo for many more exciting pictures and movies!) and I made a movie for his blog, describing the most important oceanographic instrument. We recently translated the movie from Norwegian to […]

On the structure of fresh water and salt water ice
More details on the structure of fresh water and salt water ice. Fresh water and salt water ice have very different structures as I already discussed in this post. In the image above you see that the structures are very different. Whereas fresh water ice is clear and transparent, salt water ice has a porous structure […]

Melting ice cubes – one experiment, many ways (post 3/4)
Different didactical settings in which the “ice cubes melting in fresh and salt water” experiment can be used. In part 1 and 2 of this series, I showed two different ways of using the “ice cubes melting in fresh water and salt water” experiment in lectures. Today I want to back up a little bit […]

How much salt is there in sea water?
Visualization of how much salt is actually contained in sea water. When preparing “sea water samples” for class, it is always astonishing to me how much salt I have to add for normal open-ocean salinities. Time and time again it looks like it should be way too much, but then when tasting it, it tastes […]

Properties of sea ice and fresh water ice
Sea ice and fresh water ice have distinctly different properties that can easily be investigated even in big class rooms. In “on how ice freezes from salt water” I talked a bit about how dye was rejected when I tried to produce colored ice cubes for another experiment. But even non-colored ice that were made […]

Ice cubes melting in fresh water and salt water (post 2/4)
The “ice cubes melting in fresh water and salt water” experiment the way I usually use it in class. — Edit — For an updated description of this experiment please go to this page! — Edit — You might remember the “ice cubes melting in fresh water and salt water experiment” from a couple of […]

On how ice freezes from salt water
I’ve been wondering how to best show how sea ice freezes for quite a while. Not just that it freezes, but how brine is rejected. By comparing the structure of fresh water and salt water ice, one can get an idea of how that is happening (and I’ll write a post on that after we […]

Ice cubes melting in salt water and freshwater (post 1/4)
Experiment to visualize the effects of density differences on ocean circulation. This is the first post in a series on one of my favorite in-class experiments; I have so much to say about it that we’ll have to break it up into several posts. Post 1 (this post) will present one setup of the experiment, […]

How to measure temperature, salinity and density
Three in-class experiments run in parallel. Great if you want to discuss how properties are measured and what kind of difficulties you might encounter. Temperature, salinity and density are the most important properties in physical oceanography. Measuring them with a CTD is easy. But can you, using basic household items, build instruments to measure those properties? My […]

Tasting sea water
Hands-on activity on sea water salinity In the first lecture of the “introduction to oceanography” GEOF130 course 2013, we investigated water samples from four different regions: The Mediterranean, the tropical North Atlantic, the Baltic and Arctic sea ice. Just by tasting their different salinities (40psu, 35psu, 10psu and 5psu, respectively) students figured out which of […]


