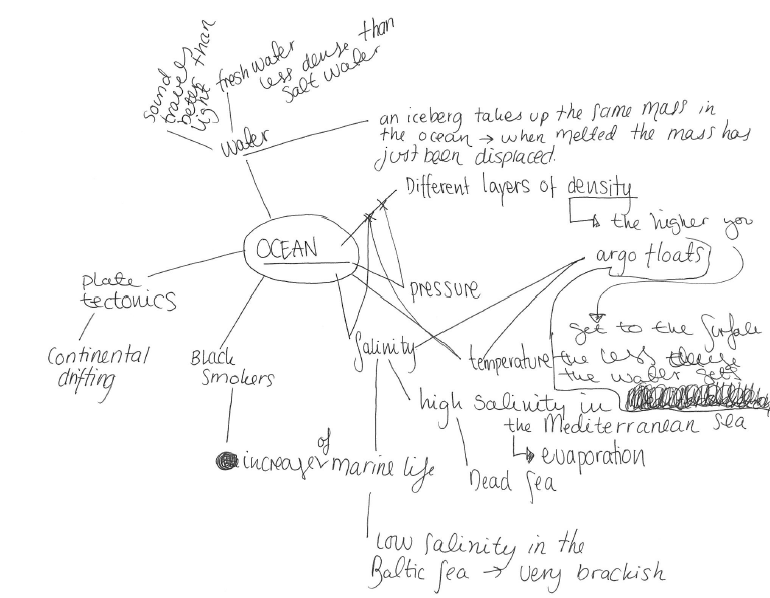
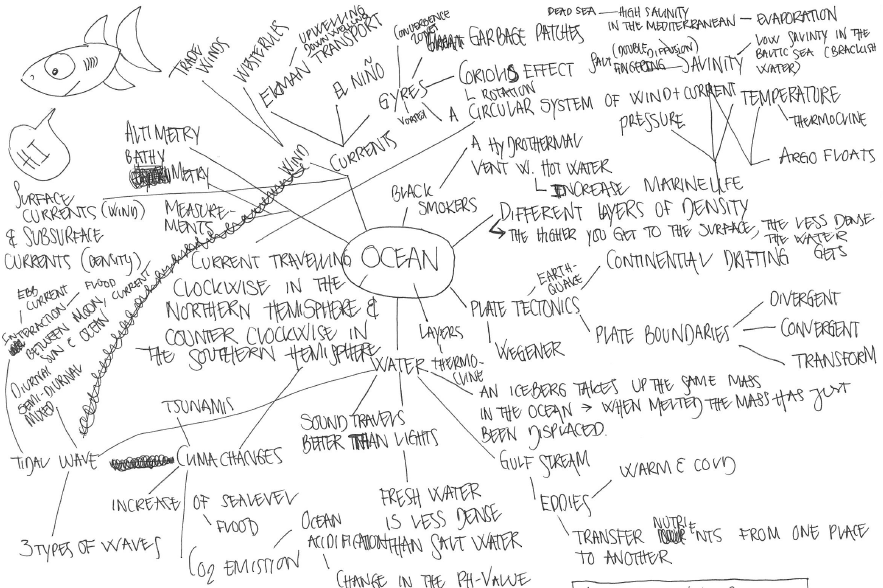
Including missing topics that students suggest
I’ve been talking about the importance of leaving room for topics that students are really interested in for a long time. Today, I want to tell you about my first experience with this: Back in 2012, in my first year teaching the “introduction to oceanography course”, a student came up to me after the first […]