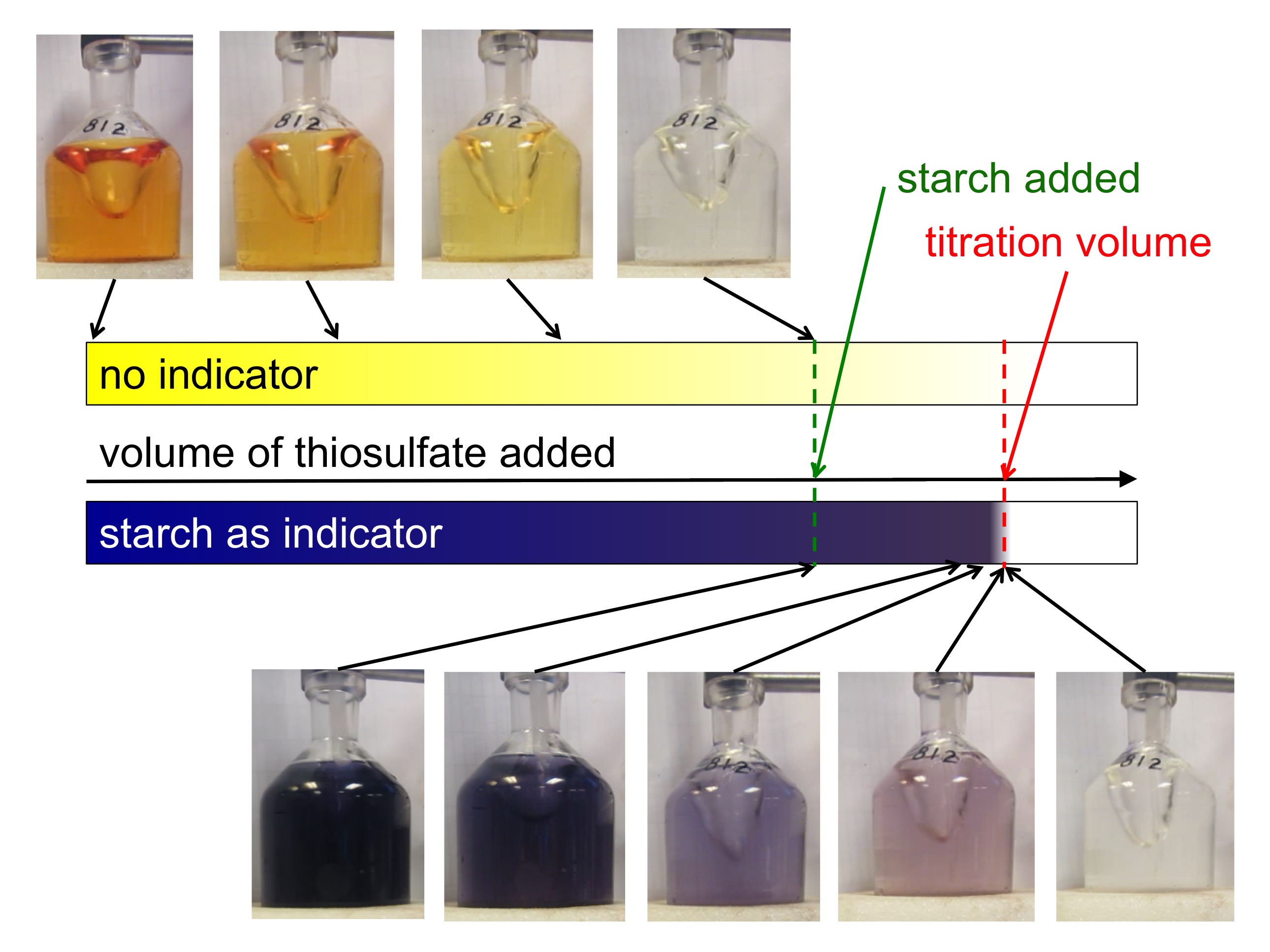
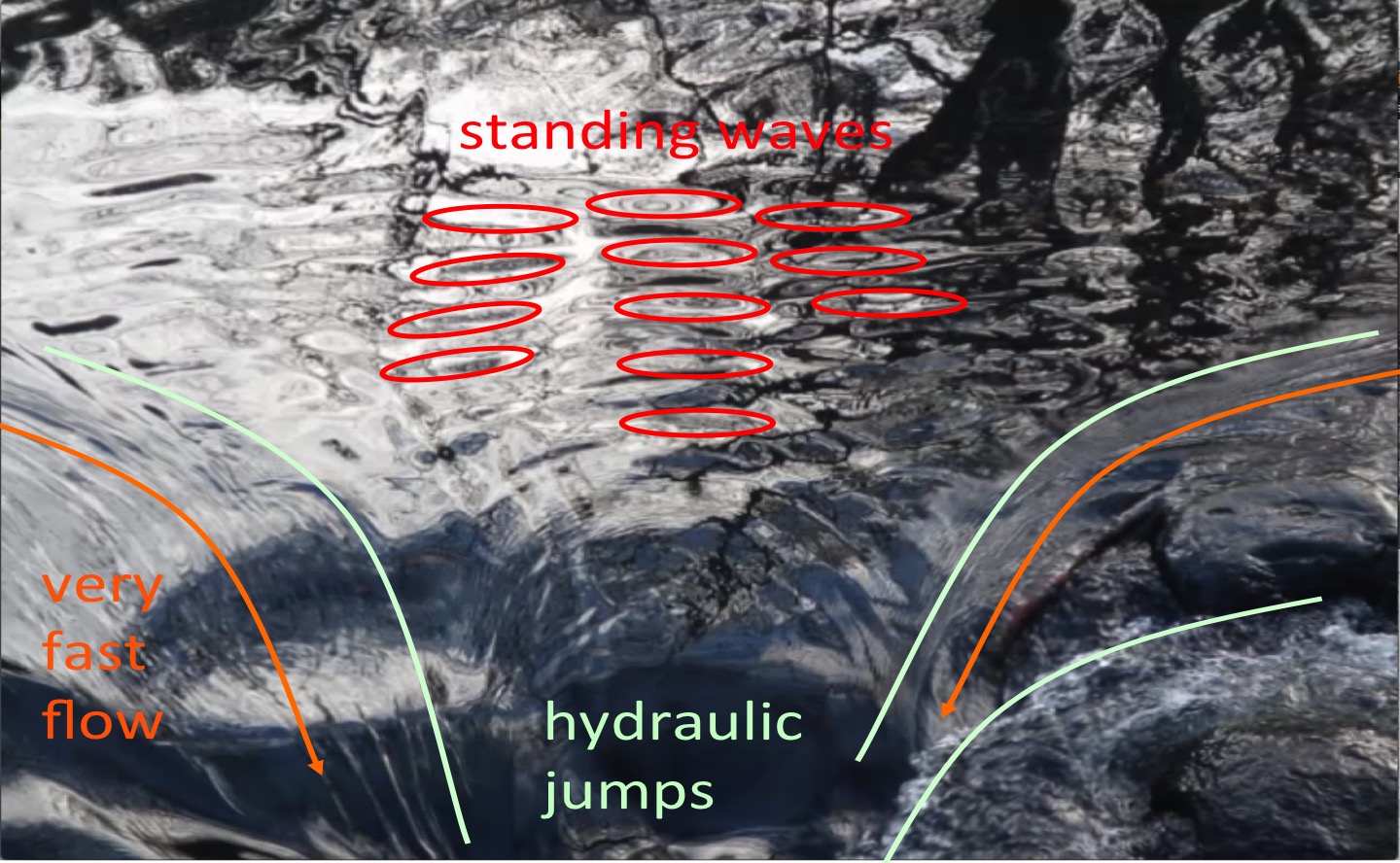
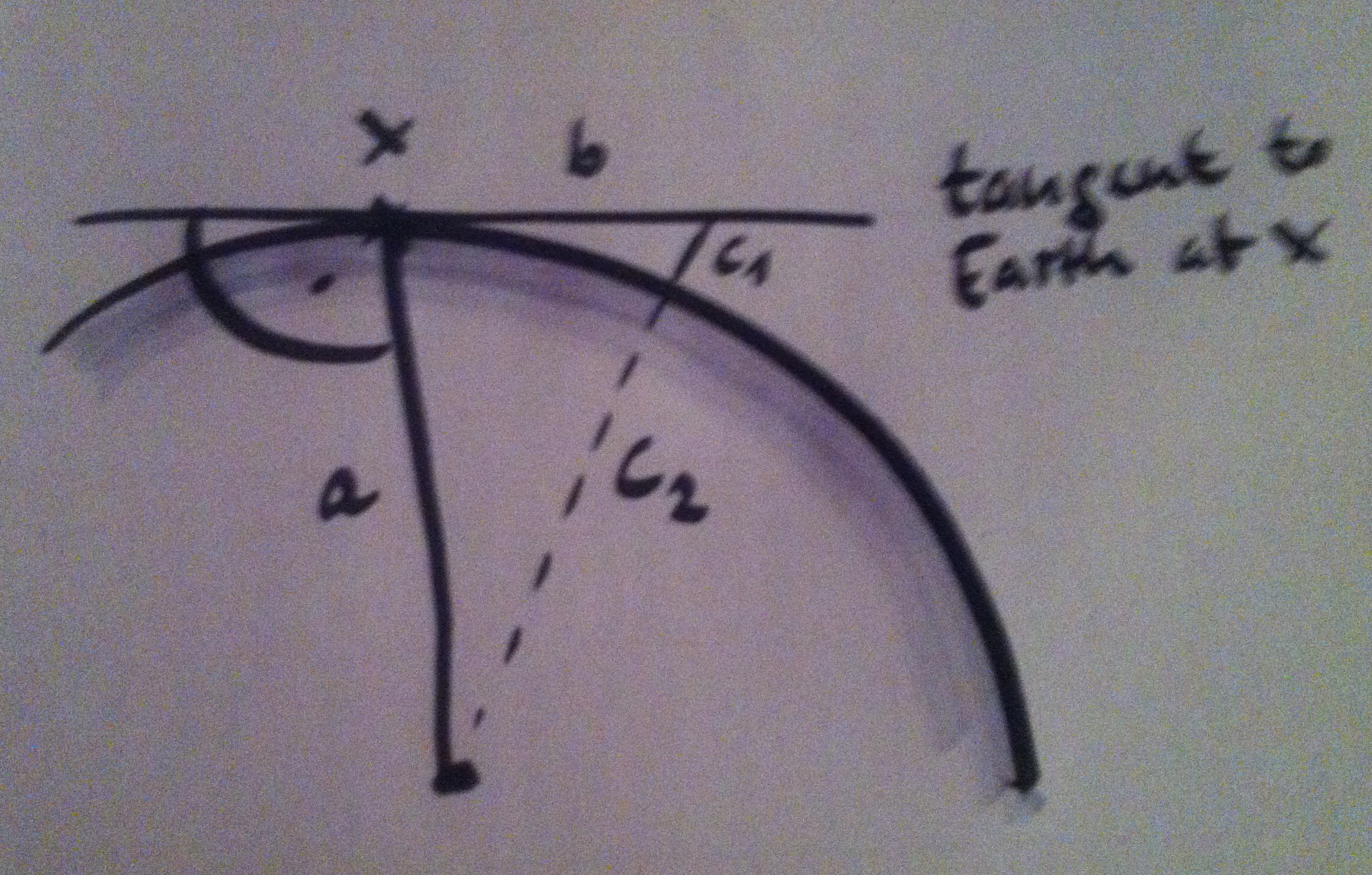
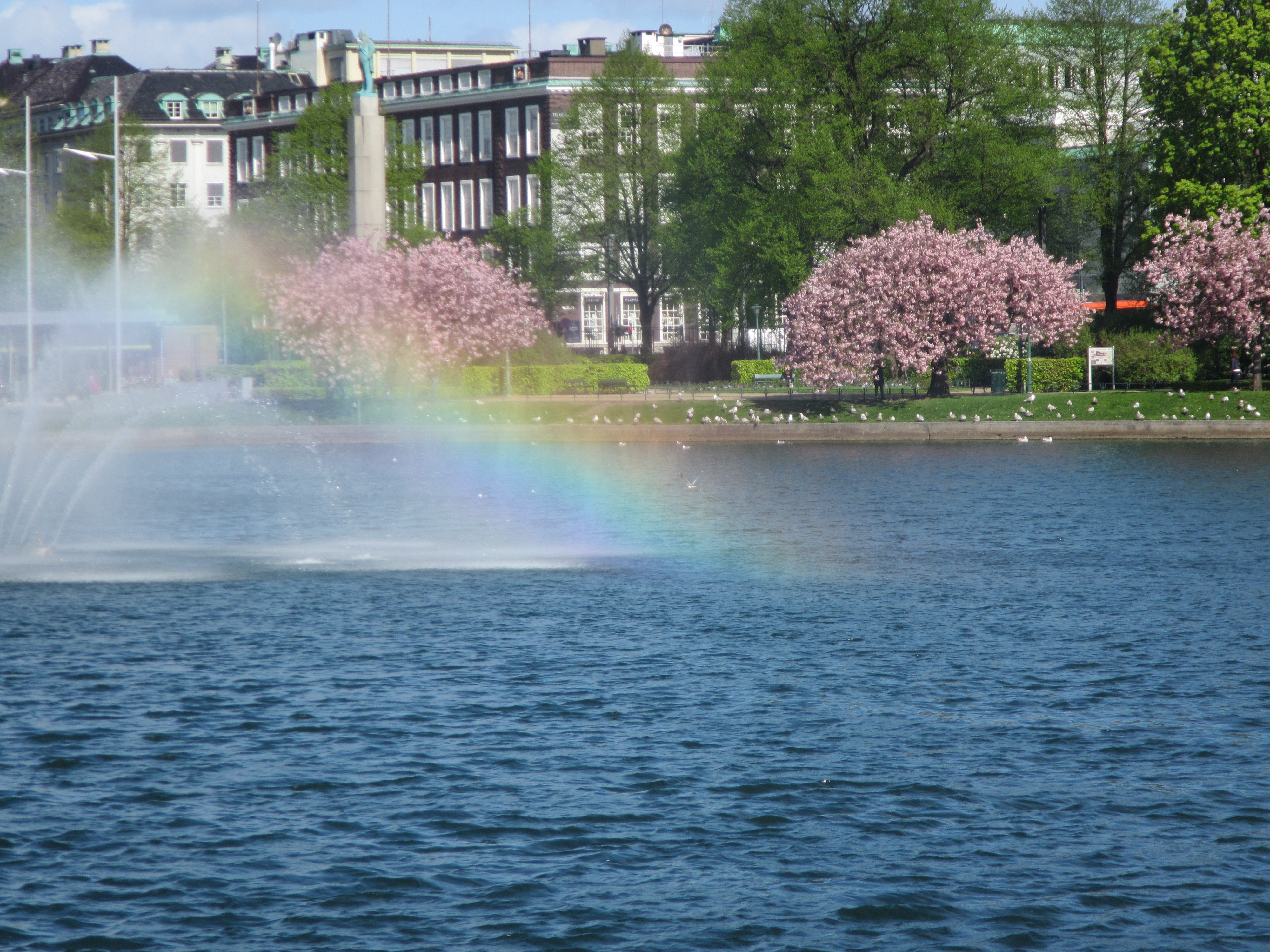Total internal reflection
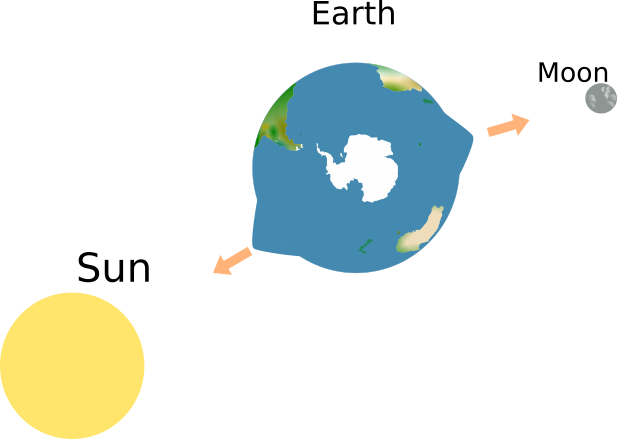
In the “static apnea” discipline in freediving, many cool pictures of athletes are taken underwater in a way that plays with the reflection of the athlete in the still water surface. This can lead to pretty spooky pictures (like the one of Victor in the top left). We do have other experiences with water, where […]