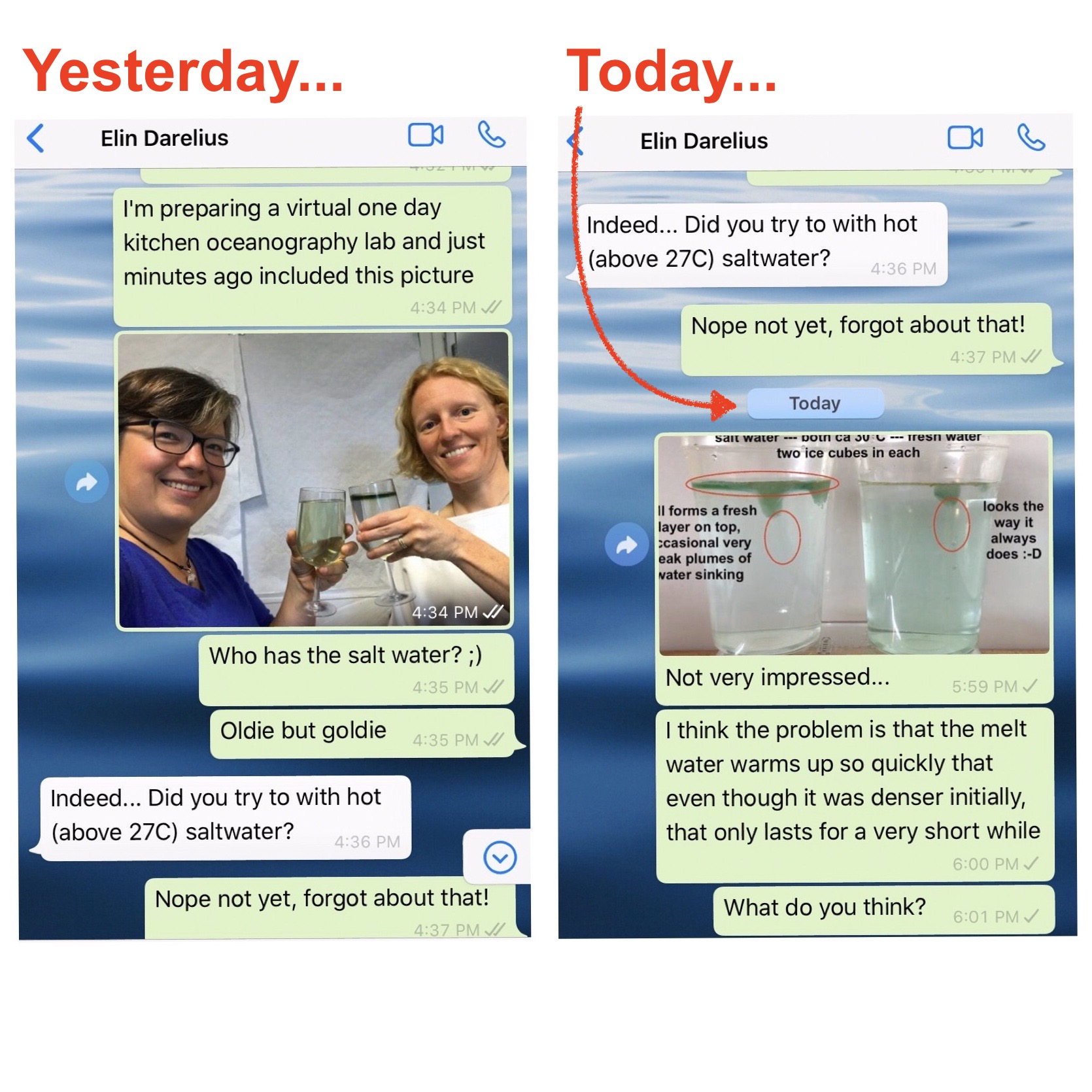
Molecular diffusion at different temperatures (involving tea bags and some convection)
I thought I had posted the picture below some time in winter already, but when I recently searched for it, I couldn’t find it. So either I didn’t post it, or I didn’t post any sensible search terms with it, in any case: It’s useless. So here we go again. Below you see two tea […]