Today’s guest blogger Jeannette and I “met” on Twitter when she reposted one of my 24 Days of #KitchenOceanography posts, saying “A friend just forwarded me a #kitchenoceanography experiment that pretty much sums up my MSc work minus all the math.”. So I — obviously — asked her to write a guest post, and here we go! Thank you, Jeannette! :-)
“Lee waves are everywhere. They lurk in your sink, form over mountains and even beneath the ocean’s surface (no doubt they’ll be found out space too).
Mountains and under-sea ridges change how a fluid (air or water) passes over it. Glider pilots in the 1930s first noted the effects of lee waves—when a glider catches a lee wave, the unpowered aircraft can climb higher and stay in the air longer adding to the fun of their flight. But since I’m an oceanographer, I’m going to focus on water.
When water pushes up and over an obstacle, it gets squeezed and speeds up. At the bottom the water slows creating a wave on the surface. How this wave moves depends on the fluid velocity and water depth which can be combined in the Froude number.
The Froude number equals the fluid velocity over the square root of gravity times water depth (note—it’s water depth, not obstacle height so it still applies to the flat landscape of your sink). By using this number, flows in dramatically different settings can be compared. For example, atmospheric flow over a mountain range can be related to water moving over a weir.
So what does the Froude number tell us?
When F is smaller than one, flow over the bump is ‘subcritical’. The resulting surface wave can travel upstream, meaning that downstream conditions affect the flow upstream. This is kind of like tossing a pebble into a flowing stream and seeing the resulting ripples move both upstream and downstream.
When F is larger than 1, flow is ‘supercritical’ meaning no surface disturbance can travel upstream. Here, ripples created by a pebble tossed in cannot overcome the speed of the water and only move downstream.
Now, back to flow over a bump (although the bump is not actually required). As subcritical water pushes over it’s squeezed as the water is now shallower but the same amount of water has to move through. This forces the water to speed up and transition to supercritical.
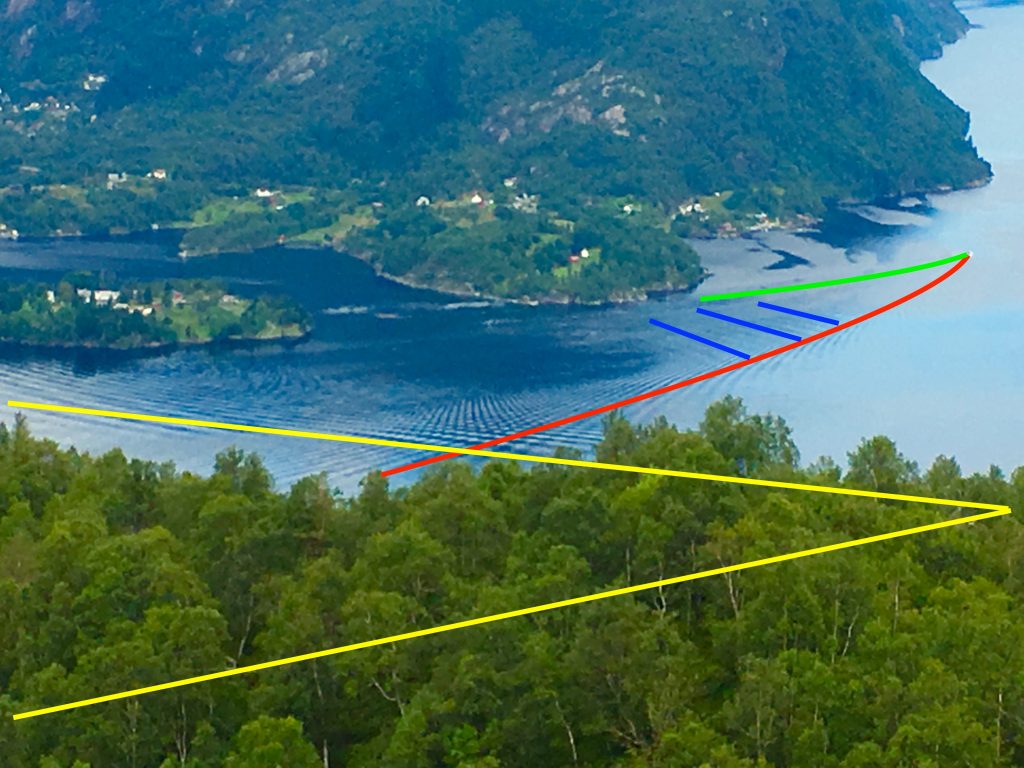
As faster water crosses over to the other side of the bump where it’s again deeper. It abruptly slows and waves form. Since the water is moving too fast to let the waves move upstream (because it is supercritical) these waves build up, forming a sudden water level increase that can stand still in the flowing water. This is called a hydraulic jump—a non-linear effect observable in a kitchen sink or in water passing over a weir.
The bigger the Froude number is, the more pronounced the jump will be. For flow speeds slightly above the critical speed, the jump forms as an undulating wave. When flow speed increases, the Froude number also increases, and the transition becomes abrupt in shape. Beneath the wave, water flow becomes chaotic in an effect called turbulence.
Because of the turbulence they create, the sea floor under a lee wave makes great habitat for critters—especially stationary filter feeders, as a buffet of tasty treats whooshes by.”