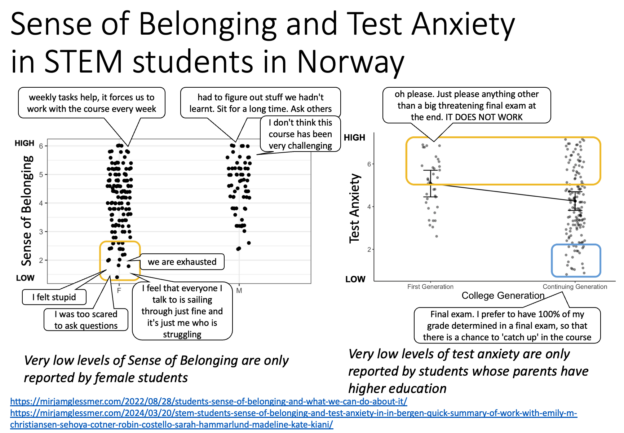
In my series of things-I-want-to-say-in-an-upcoming-workshop-but-suspect-I-might-skip-to-make-time-for-participants’-topics, here is a quick summary of work I did with Emily M. Christiansen, Sehoya Cotner, Robin Costello, Sarah Hammarlund, & Madeline Kate Kiani on STEM students’ sense of belonging in Bergen a while back. The results are very interesting, mostly because they are very similar to what we have read about for years in a US context. However, this is the first time that this type of data has been collected in a Scandinavian context, where the assumption is traditionally that gender equality is so high that there shouldn’t be any differences between male and female students’ sense of belonging (yet we see there is), and where access to education is universal, so that it shouldn’t matter whether your parents have been to university or not (yet we see a correlation with test anxiety). Becoming aware of that is really important, so that we can figure out how to address it and level the playing field (some examples of what we could do are given here).
Continue reading →